Goeser is director of nutritional research and innovation with Rock River Lab, Inc., Watertown, Wis., and adjunct assistant professor, dairy science department, University of Wisconsin-Madison. Sawyer is the laboratory manager and QA/QC director for Rock River Lab, Inc.

The variability of K in forage leads to speculation that other mineral levels may be changing as well, given variable environmental conditions. Tissue testing during the growing season, as well as understanding the environmental influence on feed quality, can help to predict the need for mineral supplements in your ration.
When balancing rations, your consultants track dietary macrominerals, including calcium, chloride, potassium, phosphorus, magnesium, sulfur and sodium. However, have you ever discussed why? Has your consultant noticed mineral levels changing in forages over the years? The answer to each should be yes.
For this discussion, we will focus on potassium (K) to exemplify and describe changes in forages, soils, plants and potentially even diets for dry or lactating cows. Realize, though, that all mineral levels may vary under your dairy's agronomic, environmental and nutritional circumstances.
K's role changes from dry to fresh
We often discuss potassium in nutrition programs, especially when evaluating DCAD (Dietary Cation Anion Difference). We feed animals potassium carbonate or other K sources, but what is actually absorbed is elemental potassium. Once potassium is solubilized, it exists as a cation (just like calcium), meaning it carries a positive ionic charge.
K enters the conversation in dry cow nutrition programs due to its role in clinical and subclinical milk fever risks at calving. Excessive dietary K intake prior to calving raises blood pH and interferes with the animal's ability to liberate calcium from bones. This situation lowers circulating blood calcium levels to less than 8 mg/dL as substantial amounts of calcium are pulled from the body to meet colostrum and milk production demands. The calcium shortage impairs muscle function (even breathing), nervous system integrity and limits performance.
Many producers focus on sourcing or purchasing feedstuffs low in potassium to help avoid this scenario, yet it is possible to go too far. K is essential and must be supplied daily and balanced to meet demands. It is actually the most concentrated mineral excreted in both colostrum and milk (more so than calcium). Body K reserves are tight, and extensive excretion coupled with low intake after calving will quickly cause deficiencies. Deficient K is referred to as hypokalemia. This condition is becoming more prevalent.
While a negative DCAD is beneficial prefresh, positive DCAD diets are beneficial to fresh and high-performing cows. As cows consume high-energy TMRs, the rumen bacteria rapidly digest carbohydrates and produce substantial amounts of volatile fatty acids (VFA). The VFA are absorbed quickly across the rumen wall into the blood and are used for energy. However, the VFA also can lower blood pH level and hinder performance. Here, supplemental K and other cations help maintain optimal blood pH levels and performance.
The first step in balancing for K and other cations is having sound measurements for all feed ingredients, which should be an X-ray or plasma-based wet chemistry measurement. At Rock River Laboratory, we have seen K levels changing over time and varying following drought periods or wet growing seasons.
Why is this? Are soil K levels variable through this period? Are plants absorbing and metabolizing K differently?
Plant K depends on water
Knowing what to expect of your growing crop is an important piece of the balancing act. The drought conditions of 2012 provided us with a unique opportunity to study the relationship between soil test and plant tissue K levels under extreme circumstances. Not surprisingly, fresh forages exhibited an identical trend to K levels found in soils; both were lower than would be found in a typical growing season.
The soil/plant dynamic is extremely dependent on water. Just as in animals, plants take up K in the ionic form, K+. In soil, this form of potassium is known as "solution K," as it resides in the soil-water solution. Without water in the soil to dissolve K into this usable form, the K remains bound in various compounds and spaces that are simply unavailable to the plant.
Any solution K that may be in the soil requires an uninterrupted chain of water-filled pores in order to flow toward the plant's roots. As soil pores dry out, these pathways to the root become fewer and fewer. This means that diffusion, the primary delivery method of K to the plant root, is greatly inhibited. To further complicate the story, plant roots will move deeper into the subsoil as they search for water.
Nutrient stratification studies have shown a difference in chisel-plowed soils as great as 100 parts per million (ppm) between soil test K in the top 2 inches and soil test K at an 8-inch depth. The combined impact of deep roots and limited mobility means that surface-applied fertilizers may have little impact on crop production. Irrigation, however, may be able to free up soil K for use by the plant.
Potassium is still in the soil under drought conditions, it's just not available to the plants. The good news is that it will become available again when soil moisture returns to normal.
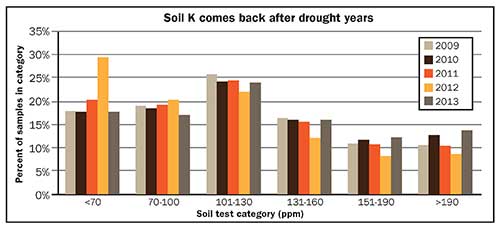
The figure shows the percentage of Wisconsin soil samples in each of six soil test K categories from 2009 to 2013. Notice that in 2012 the percentage of samples with soil test K levels less than 70 ppm rose nearly 10 percent. In 2013, when soil moisture levels returned to normal, the incidence of samples in this category returned to predrought levels. This tells us that drought-induced soil K deficiencies are temporary.
Work with your nutrition and agronomic consulting team to better understand these dynamic interactions. Consider wet chemistry mineral measures for all diets to ensure usable mineral requirements are met.
Click here to return to the Crops & Forages E-Sources
140510_334

When balancing rations, your consultants track dietary macrominerals, including calcium, chloride, potassium, phosphorus, magnesium, sulfur and sodium. However, have you ever discussed why? Has your consultant noticed mineral levels changing in forages over the years? The answer to each should be yes.
For this discussion, we will focus on potassium (K) to exemplify and describe changes in forages, soils, plants and potentially even diets for dry or lactating cows. Realize, though, that all mineral levels may vary under your dairy's agronomic, environmental and nutritional circumstances.
K's role changes from dry to fresh
We often discuss potassium in nutrition programs, especially when evaluating DCAD (Dietary Cation Anion Difference). We feed animals potassium carbonate or other K sources, but what is actually absorbed is elemental potassium. Once potassium is solubilized, it exists as a cation (just like calcium), meaning it carries a positive ionic charge.
K enters the conversation in dry cow nutrition programs due to its role in clinical and subclinical milk fever risks at calving. Excessive dietary K intake prior to calving raises blood pH and interferes with the animal's ability to liberate calcium from bones. This situation lowers circulating blood calcium levels to less than 8 mg/dL as substantial amounts of calcium are pulled from the body to meet colostrum and milk production demands. The calcium shortage impairs muscle function (even breathing), nervous system integrity and limits performance.
Many producers focus on sourcing or purchasing feedstuffs low in potassium to help avoid this scenario, yet it is possible to go too far. K is essential and must be supplied daily and balanced to meet demands. It is actually the most concentrated mineral excreted in both colostrum and milk (more so than calcium). Body K reserves are tight, and extensive excretion coupled with low intake after calving will quickly cause deficiencies. Deficient K is referred to as hypokalemia. This condition is becoming more prevalent.
While a negative DCAD is beneficial prefresh, positive DCAD diets are beneficial to fresh and high-performing cows. As cows consume high-energy TMRs, the rumen bacteria rapidly digest carbohydrates and produce substantial amounts of volatile fatty acids (VFA). The VFA are absorbed quickly across the rumen wall into the blood and are used for energy. However, the VFA also can lower blood pH level and hinder performance. Here, supplemental K and other cations help maintain optimal blood pH levels and performance.
The first step in balancing for K and other cations is having sound measurements for all feed ingredients, which should be an X-ray or plasma-based wet chemistry measurement. At Rock River Laboratory, we have seen K levels changing over time and varying following drought periods or wet growing seasons.
Why is this? Are soil K levels variable through this period? Are plants absorbing and metabolizing K differently?
Plant K depends on water
Knowing what to expect of your growing crop is an important piece of the balancing act. The drought conditions of 2012 provided us with a unique opportunity to study the relationship between soil test and plant tissue K levels under extreme circumstances. Not surprisingly, fresh forages exhibited an identical trend to K levels found in soils; both were lower than would be found in a typical growing season.
The soil/plant dynamic is extremely dependent on water. Just as in animals, plants take up K in the ionic form, K+. In soil, this form of potassium is known as "solution K," as it resides in the soil-water solution. Without water in the soil to dissolve K into this usable form, the K remains bound in various compounds and spaces that are simply unavailable to the plant.
Any solution K that may be in the soil requires an uninterrupted chain of water-filled pores in order to flow toward the plant's roots. As soil pores dry out, these pathways to the root become fewer and fewer. This means that diffusion, the primary delivery method of K to the plant root, is greatly inhibited. To further complicate the story, plant roots will move deeper into the subsoil as they search for water.
Nutrient stratification studies have shown a difference in chisel-plowed soils as great as 100 parts per million (ppm) between soil test K in the top 2 inches and soil test K at an 8-inch depth. The combined impact of deep roots and limited mobility means that surface-applied fertilizers may have little impact on crop production. Irrigation, however, may be able to free up soil K for use by the plant.
Potassium is still in the soil under drought conditions, it's just not available to the plants. The good news is that it will become available again when soil moisture returns to normal.

The figure shows the percentage of Wisconsin soil samples in each of six soil test K categories from 2009 to 2013. Notice that in 2012 the percentage of samples with soil test K levels less than 70 ppm rose nearly 10 percent. In 2013, when soil moisture levels returned to normal, the incidence of samples in this category returned to predrought levels. This tells us that drought-induced soil K deficiencies are temporary.
Work with your nutrition and agronomic consulting team to better understand these dynamic interactions. Consider wet chemistry mineral measures for all diets to ensure usable mineral requirements are met.
140510_334











